Pulling teeth: When 20th Century Regulation Meets 21st Century Products
Enhanced Internet connectivity has transformed many markets over the years. From cars and video games, to refrigerators and windows (not the operating system, but the transparent glass ones in your house), it seems that fewer and fewer “traditional” markets and industries can resist the benefits of Internet connectivity. This phenomenon is empowering a new generation of entrepreneurs to transform everyday household devices in ways that the current market leaders would never dream about. This also means that more and more regulatory agencies, without a history of dealing with the Internet or cutting edge technology, will have their say in how older products integrate new features.
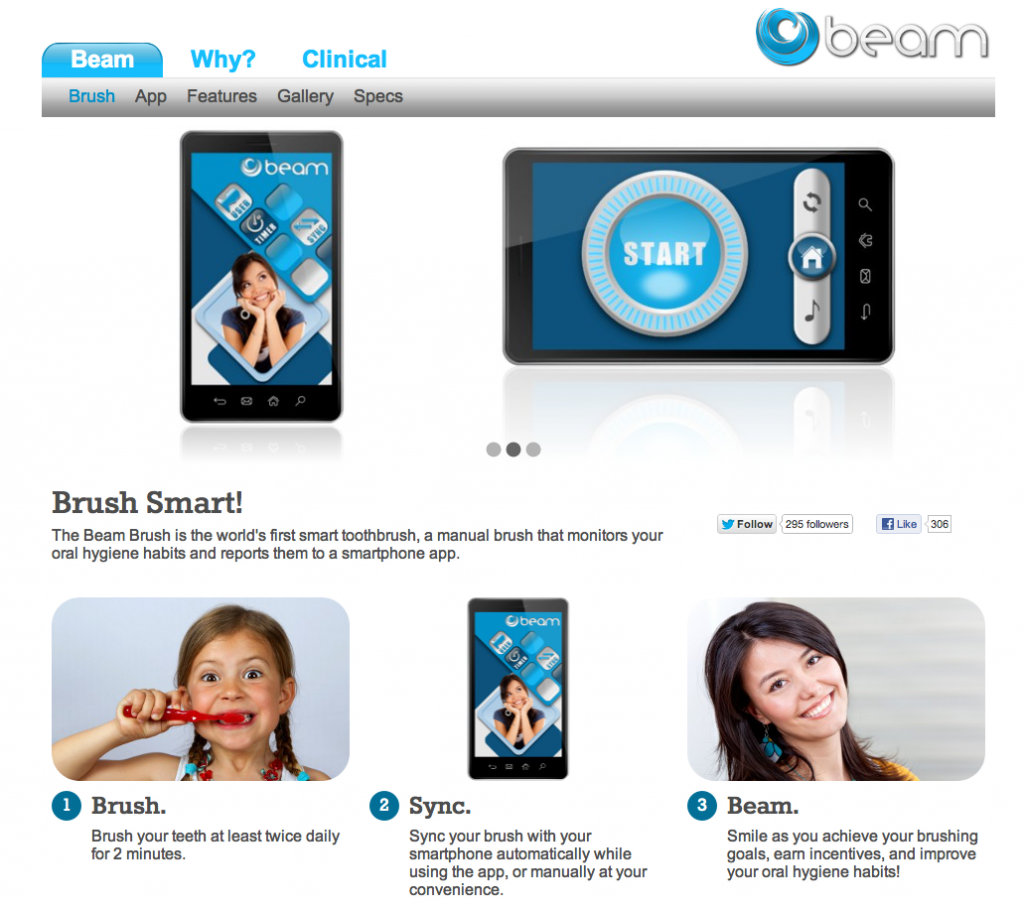
Stacey Higginbotham of GigaOm brings us a story about Alex X. Frommeyer, an entrepreneur and the CEO of Beam Industries, who recently introduced a bluetooth enabled electric toothbrush.
The first generation toothbrush may not appeal to those seeking the ultimate in data on their dental hygiene, as it will consist mostly of letting you track how long and when you brushed your teeth. To jazz up the experience, Frommeyer will let the app play songs and may incorporate a social element or game element into the process….
…Future iterations will also be able to show the user how long they spent in certain areas of the mouth. The sensor works via contact with the mouth, so it registers when the brush is scrubbing your teeth. I’d love for the app to become almost a reward system and prompt for good brushing, so when my daughter (or I) is brushing, the app could play a video or something fun, but if the brushing slacked off or spent too long in one place, the video stopped playing. I’m sure there are those of you who are scoffing at the idea that one can’t just suck it up for the two minutes it takes to brush your teeth, but I’m on board. After all, I already read a book or magazine while brushing, so I’m happy to be entertained while performing a mundane task.
Although the capabilities of the toothbrush are not revolutionary, the future uses of an interconnected toothbrush could be pretty exciting… well, as exciting as a dental hygiene experience could be anyway.
The promise of a connected toothbrush isn’t just a better brushing experience in Frommeyer’s eyes. He’d like patients to share that data with their dentists and hygienists. On an individual level that means better patient care (and you can still lie about flossing if you want), and on a macro level it could mean sharing aggregated data from millions of Beam Brush users, and finding patterns or shifts in how tooth decay occurs.
However, the FDA, wary of the perils posed by a market of cyber toothbrushes, is ensuring that even the simple step of giving a toothbrush bluetooth connectivity can not escape a drawn out regulatory approval process.
Frommeyer has long had plans to create medical products geared at more effective data collection, but started with dentistry because it’s supposedly less regulated by the FDA. In fact, he registered the toothbrush under the impression that it wouldn’t need FDA approval after they had taken a look at the design. That way, his product could begin to address the myriad of challenges associated with big data collection in the medical field without first having to clear the FDA’s bureaucratic hurdles.
No dice. The FDA phoned Frommeyer to tell him that his invention was a “new class of toothbrush” and that it would have to be reviewed in order to receive the agency’s approval. Frommeyer said that he thought they had a weak argument, but didn’t have a choice other than to comply. Four months later, the toothbrush is ready to hit the market. And Frommeyer is lucky — four months is a relatively short time compared to the FDA’s norm. That said, in a dynamic industry like mobile apps, regulatory overkill can be incredibly detrimental to innovation. Yes, our teeth are important and nobody wants to take an electric shock to the mouth, but overreactions to inventions like Frommeyer’s will only hamper the motivation for entrepreneurs to enter the vital field of medical IT. As the GigaOm story points out, this anecdote is troubling largely for what it means for more ambitious products in the medical space:
As more startups seek to enter the medical IT space with connected devices, apps or new services, the looming threat of the FDA’s famously lengthy approval process is something most want to avoid. Corey Ackerman, president of Happtique, once shared an anecdote with me about a doctor who got approval for a medical app that was certified on an early version of the Android operating system only to realize that technically upgrading the app to later OSes would void the approval.
To give the FDA credit, they are aware of this phenomenon. However, their approach to it seems less about nurturing innovation and more about making sure it remains under their control. As the FDA’s new draft regulations for mobile applications state:
This narrowly-tailored approach focuses on a subset of mobile apps that either have traditionally been considered medical devices or affect the performance or functionality of a currently regulated medical device. The FDA believes that this subset of mobile apps poses the same or similar potential risk to the public health as currently regulated devices if they fail to function as intended. Using mobile or other innovative platforms along with a mobile medical app to perform medical device functions does not necessarily change the intended use or the risk to patients if the device fails to operate properly.
As the draft guidelines make clear, mobile apps can be regulated if they affect the performance of a “currently regulated medical device.” This seems particularly backwards looking (it focuses on products that the FDA traditionally regulates and not what the mobile app actually does). In this exercise of bureaucratic tautology, the FDA is basically saying: “We are regulating what we regulate because we regulate it” (Or as Oscar Wilde said, “Bureaucracy is expanding to meet the needs of the expanding bureaucracy”). This is inherently at odds with innovators, who see old problems and think of new ways to solve them.
Although the FDA pays lip-service to the “narrowly-tailored” approach to regulation of mobile apps, adding capabilities to time your brushing session and play music during it seem rather innocuous. Even if they don’t function “properly”–which appears to be the stated goal of the regulation–its seems like such a scenario can be dealt with by the FTC’s truth in advertising regulations.
Regulators, particularly in the healthcare space, should be focused on encouraging innovation–not on forcing it into the current framework of regulation. Saddling a new startup with massive regulatory review, especially when it is unnecessary, is a surefire way to stop many innovators from bringing their products to market. (In this case, Mr. Frommeyor decided to start with toothbrushes even though he had bigger ideas because he figured the regulatory barriers would be minimal.) As I discussed in the case of Airbnb and Uber, regulators–or the legislators that oversee them–should avoid the temptation to bring new services under old regulatory paradigms that not only make no sense, but actually work against the outcomes that they were designed to achieve.
With healthcare costs rising astronomically (while not necessarily translating into better health outcomes), the health care industry “screams for disruption,” as Clayton Christensen himself has recently noted. If entrepreneurs can integrate a toothbrush with the Internet, then surely regulators can evolve from 20th century notions of regulation.








